I am interested in mechanisms that have created and maintained species diversity of cichlid fish in Lake Tanganyika (2 to 9). In Japan, I am studying food habits of great cormorant (1).
| 9. | Mechanism maintaining colour dimorphism of males in Cyprichromis species (in progress) |
| 8. | Invisible pair bonds |
| 7. | Natural selection restricting body size |
| 6. | Lunar synchronization of spawning |
| 5. | Divergent natural selection |
| 4. | Disassortative mating |
| 3. | Identirication System for Tanganyikan Cichlids |
| 2. | Taxonomy and systematics of cichlid fish from Lake Tanganyika |
| 1. | Food habits of great cormorant |
8. Invisible pair bonds
|
This study is the first to detect pair bonds in animals in which physical proximity has not been observed. The following is the abstract of Takahashi et al. (2011): A focus on pair bonds between males and females is fundamental to study the evolution of social organization. Because pair bonds are generally identified from direct observations of pairs that maintain physical proximity, pair bonds may have been overlooked in animals that do not exhibit such visible pairs. The Lake Tanganyika cichlid fish Xenotilapia rotundiventralis forms schools that consist of mouthbrooding and nonbrooding adults in mid-water, and visible pairs are not recognized. A previous study suggested that mouthbrooding females transfer fractions of the young to males when the young become large. However, it remains a mystery whether the mating pairs maintain pair bonds so that the females can transfer the young to their mates. To answer this question, we conducted a parentage analysis using 10 microsatellite markers. The analysis showed that the mouthbrooding adults were most likely genetic fathers and mothers of the young in their mouths. This finding suggests that the female-to-male shift of young takes place between mating partners, and thus the mating pairs maintain pair bonds at least until the shift of young. The present study is the first to detect pair bonds in animals in which physical proximity has not been observed. Takahashi T et al. (published online before print) Biology Letters |
 A school of Xenotilapia rotundiventralis. Some individuals in the photo appear to be mouth-brooding. |
7. Natural selection restricting body size
|
Body size reaults from a balance between selection favouring large body size and counterbalancing selection favouring small body size.
Sexual selection in males and fecundity selection in females are widely agreed to be the major evolutionary forces in males and fecundity selection in females are widely agreed to be the major evolutionary forces that favour large body size.
However, evidence of counterbalancing seletion limiting body size is still sparse and requires more research. (from Introduction) The present study provides support for the idea that male and female body sizes have been limited by different ecological pressures (predation on adult fish in males, predation on eggs in females), which has not been reported previously in any animal. (from abstract) Takahashi T et al. (2012) Hydrobiologia 684:35-44 |
 The large fish is a male, and the small fish is a female. A male and a female rarely share the same shell. 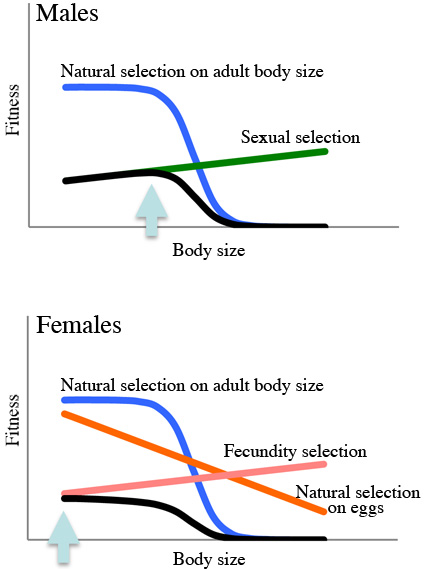 These graphs illustrate expected fitness of males and females. Adaptive body size is larger in the males. |
6. Lunar synchronization of spawning
|
Lunar cyclic spawning has been reported in substrate-brooding cichlids from Lake Tanganyika, although it is rare in the other freshwater fishes.
This phenomenon may reduce the risk of egg predation because carnivorous catfish is active during new moon in this lake.
Therefore, the degree of lunar synchronization of spawning may be related to a strategy that decreases predation on the brood.
In this study, I examined the periodic change of ovary development in the normal and dwarf morphs of Telmatochromis temporalis. As a result, the degree of lunar synchronization of ovary development was much lower in the dwarf morph than the normal morph. The dwarf morph spawns eggs inside empty snail shells, whereas the normal morph spawns in spaces under stones. The spaces inside shells are small, and probably highly effective for predator avoidance. Mothers of the dwarf morph may care for the brood safely in the small, safe spaces without synchronizing spawning with lunar cycles. Takahashi T (2010) Hydrobiologia 644:139-143 |
 The dwarf morph spawns eggs inside shells close to the apices (this shell was brocken in order to show the eggs: original size of this shell is about 4 cm in height) |
5. Divergent natural selection
|
I found a novel example of divergent natural selection form nature.
The following is the abstract of Takahashi et al. (2009): Divergent natural selection is thought to play a vital role in speciation, but clear, measurable examples from nature are still few. Among the many possible sources of divergent natural selection, predation pressure may be important because predators are ubiquitous in food webs. Here, we show evidence for divergent natural selection in a Lake Tanganyika cichlid, Telmatochromis temporalis, which uses burrows under stones or empty snail shells as shelters. This species contains normal and dwarf morphs at several localities. The normal morph inhabits rocky shorelines, whereas the dwarf morph invariably inhabits shell beds, where empty snail shells densely cover the lake bottom. Genetic evidence suggested that the dwarf morph evolved independently from the normal morph at two areas, and morphological analysis and evaluation of habitat structure revealed that the body sizes of morphs closely matched the available shelter sizes in their habitats. These findings suggest that the two morphs repeatedly evolved through divergent natural selection associated with the strategy for sheltering from predators. Takahashi T et al. (2009) Mol Ecol 18:3110-3119 Takahashi T (2004) J Fish Biol 65:419-435 |
 A mature male of the dwarf morph with its shelter of an empty snail shell (left) and a mature male of the normal morph (right)  Dwarf morph in a shell |
4. Disassortative mating
|
This study showed that the scale-eating cichlid fish Perissodus microlepis exhibited disassortative mating, in which reproductive pairings between lefties and righties occurred at higher than expected frequency.
Disassortative mating is thought to function as inbreeding-avoidance mechanism in humans and mice that have highly polymorphoic heritable phenotypes (e.g. odour).
However, disassortative mating in this fish probably does not function in inbreeding avoidance because this fish has only two morphs.
To find adaptive significance of disassortative mating in P. microlepis may give a hint to resolve mystery regarding the other, well-known disassortative mating between two morphs "sex."
Takahashi T and Hori M (2008) Biol Lett 4:497-499 |
 Guarding pair of Perissodus microlepis (upper: female, lower: male) |
3. Identirication System for Tanganyikan Cichlids
|
The taxonomic identification of organisms plays a fundamental role in many fields of biology.
Identirication, however, often requires a large amount of taxonomic knowledge about organisms.
The Identirication System for Tanganyikan Cichlids (ISTC) help you identify the species of Tanganyikan cichlids.
You have only to count five meristics, and observe three characters.
Takahashi T et al. (2007) Ichthyol Res 54:399-403 |
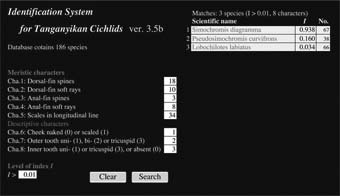 Window of ISTC |
2. Taxonomy and systematics of cichlid fish from Lake Tanganyika
|
Lake Tanganyika is one of the three great lakes of east Africa.
This lake is extremely large with dimensions 650 km in length, 50 km in width, and 1,470 m in depth at the deepest point,
and is shared by Democratic Republic of the Congo, Zambia, Tanzania, and Burundi.
This lake harbours an extraordinarily rich fauna and flora, making a characteristically endemic ecosystem.
For example, 58% of 1096 animal species, which are known from this lake, are endemic to the lake (Coulter 1994, Arch. Hydrobiol. Beih. Ergebn. Limnol. 44:13-18).
However, recent human activities appear to have changed this valuable ecosystem:
1) global warming that decreases deep-water nutrient upwelling (e.g. O'Reilly et al. 2003, Nature 424:766-768);
2) watershed deforestration that increases sediment accumulation rates (Cohen et al. 2005, J. Paleolimnol. 34:125-145);
3) overfishing on commercial fishes for food and hobby (Sturmbauer 2008, Hydrobiologia 615:95-101);
and 4) release of fish (my observation at Mpulungu).
Cichlid fish from Lake Tanganyika are one of the most spectacular examples of adaptive radiation within any vertebrate family. Approximately 200 species have been described, and more are yet to be described. Dr. Snoeks (2000, Adv. Ecol. Res. 31:17-38) estimated that about 50 species awaits description. In order to understand its biodiversity, I am studying these fishes in terms of taxonomy and systematics. Takahashi T (2008) J Fish Biol 72:603-613 Takahashi T and Hori M (2006) J Fish Biol 68B:174-192 Takahashi T et al. (2006) J Fish Biol 68A:56-67 Takahashi T (2004) Ichthyol Res 51:1-4 Takahashi T and Nakaya K (2003) Copeia 2003:824-832 Takahashi T (2003) Ichthyol Res 50:367-382 Takahashi T (2003) Species Divers 8:1-26 Takahashi T (2003) Ichthyol Res 50:36-47 Takahashi T et al. (2002) Copeia 2002:1029-1036 Takahashi T and Nakaya K (1999) Copeia 1999:101-106 Takahashi T and Nakaya K (1998) Ichthyol Res 45:413-418 Takahashi T and Nakaya K (1997) Ichthyol Res 44:335-346 Takahashi T et al. (1997) Ichthyol Res 44:109-117 |
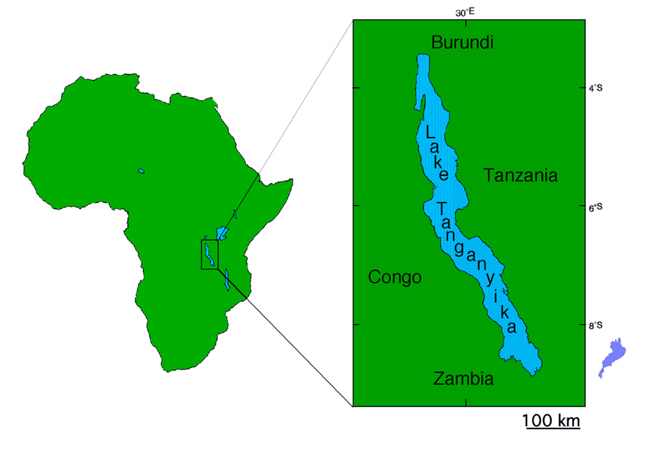 Lake Tanganyika is much larger than Lake Biwa (lower right), which is the largest lake in Japan .jpg) Number of described species of cichlid fish from Lake Tanganyika |
1. Food habits of great cormorant
|
The recent increase in numbers of individuals of a piscivorous bird, the great cormorant
Phalacrocorax carbo hanedae, around Lake Biwa has affected the commercial catch of ayu Plecoglossus
altivelis altivelis. The food habits of the bird were studied based on 66 stomachs in 1998 and
2001–2002, and a seasonal variation in food contents was observed. The proportion of ayu in stomachs
increased after April, and became most dominant during July and August (0.62 in proportion of
weight, 0.85 in frequency of occurrence). In contrast, ayu was not found in the stomachs after late
October. The increase of ayu in individuals and density in the surface layer of the pelagic zone of the
lake during spring was considered as a trigger for the change of the food habits of the birds. The feeding
impact of great cormorant on ayu in Lake Biwa was estimated to be approximately 46 t during April
to June, and 310 t in July and August. During the main period for the commercial ayu catch (November
to the following June), the estimated mass consumed by the bird is much smaller in comparison with
the commercial catch. (Abstract of Takahashi et al. 2006)
Takahashi T et al. (2006) Fish Sci 72:477-484 Takahashi T et al. (2003) Nippon Suisan Gakkaishi 69:643-645 Takahashi T et al. (2002) Nippon Suisan Gakkaishi 68:576-578 |
.jpg) Proportion of weight and frequency of ayu found in stomachs of great cormorant around Lake Biwa .jpg) Life cycle of landlocked ayu in the Lake Biwa system |
Home